Medicine Trolly Suppliers > Sechrist Industries, Inc.
Sechrist Industries, Inc.
Click Here To EMAIL INQUIRYPhone: +1-(714)-579-8400, 800-SECHRIST | Fax: +1-(714)-579-0814
Address: 4225 E La Palma Ave, Anaheim, California 92807, USA
E-Mail:
Web: https://sechristusa.com
Inquiry
Product Videos: Hide/Show >>
Sechrist Industries, Inc. primarily supplies hyperbaric and respiratory equipment in over 172 countries for the hyperbaric therapy market. Our respiratory products incudes critical care neonatal/infant/pediatric ventilators, monitoring equipment and low pressure gas mixers and patient gurneys. We are accredited with ISO 13485:2003 certification.
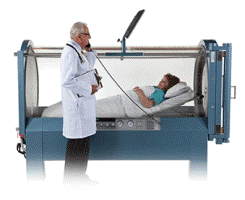
Our Monoplace Hyperbaric Oxygen Chamber remains the largest diameter chamber on the marketplace. In 2017, Sechrist offered a dual-use Hyperbaric Oxygen & ICU Chamber for the Veterinary marketplace. Shortly after Sechrist introduced the new E-Gurney? Electric Wheeled Stretcher. The E-Gurney will allow caregivers to raise, lower, brake the stretcher with the touch of an electronic foot pedal.
The company was founded in 1973, has revenues of USD 10-25 Million, has ~90 employees and is ISO, CE certified.
FDA Registration Number: 2020676
Hyperbaric Oxygen Chambers | Hyperbaric Gurneys | Air and Oxygen Gas Blenders | Exhilation Valves | Infant Ventilators | Hyperbaric Positioning Pads | Hyperbaric Chamber Mattress
Image Catalog:
| • Air/Oxygen Gas Blenders |
| • Anesthesia Inhalation Breathing Gases Mixer (FDA Code: BZR / 868.5330) A breathing gas mixer is a device intended for use in conjunction with a respiratory support apparatus to control the mixing of gases that are to be breathed by a patient. |
| • Blenders |
| • Cardiopulmonary Bypass Oxygenator (FDA Code: DTZ / 870.4350) A cardiopulmonary bypass oxygenator is a device used to exchange gases between blood and a gaseous environment to satisfy the gas exchange needs of a patient during open-heart surgery. |
| • Clinical Acrylic Monoplace Hyperbaric Chamber |
| • Continuous Ventilator (FDA Code: CBK / 868.5895) A continuous ventilator (respirator) is a device intended to mechanically control or assist patient breathing by delivering a predetermined percentage of oxygen in the breathing gas. Adult, pediatric, and neonatal ventilators are included in this generic type of device. |
| • Cutaneous Carbon-Dioxide Monitor (FDA Code: LKD / 868.2480) A cutaneous carbon dioxide (PcCO2) monitor is a noninvasive heated sensor and a pH-sensitive glass electrode placed on a patient's skin, which is intended to monitor relative changes in a hemodynamically stable patient's cutaneous carbon dioxide tension as an adjunct to arterial carbon dioxide tension measurement. |
| • Electronic Gas Blenders |
| • Exhilation Valves |
| • Fluid Delivery Tubing (FDA Code: FPK / 880.5440) An intravascular administration set is a device used to administer fluids from a container to a patient's vascular system through a needle or catheter inserted into a vein. The device may include the needle or catheter, tubing, a flow regulator, a drip chamber, an infusion line filter, an I.V. set stopcock, fluid delivery tubing, connectors between parts of the set, a side tube with a cap to serve as an injection site, and a hollow spike to penetrate and connect the tubing to an I.V. bag or other infusion fluid container. |
| • Hyperbaric Chamber (FDA Code: CBF / 868.5470) A hyperbaric chamber is a device that is intended to increase the environmental oxygen pressure to promote the movement of oxygen from the environment to a patient's tissue by means of pressurization that is greater than atmospheric pressure. This device does not include topical oxygen chambers for extremities (878.5650). |
| • Hyperbaric Chamber Mattress |
| • Hyperbaric Gurneys |
| • Hyperbaric Oxygen Chamber |
| • Hyperbaric Positioning Pads |
| • Hyperbaric Therapy |
| • Infant Transport Ventilator |
| • Infant Ventilator |
| • Mechanical Alarms |
| • Mechanical Neonatal Ventilation |
| • Mechanical Noninvasive Ventilators |
| • Mechanical Ventilation Circuits |
| • Mechanical Ventilation, Peep Valves |
| • Mechanical Ventilators |
| • Neonatal Ventilators |
| • Non-Rebreathing Valve (FDA Code: CBP / 868.5870) A nonrebreathing valve is a one-way valve that directs breathing gas flow to the patient and vents exhaled gases into the atmosphere. |
| • Pediatric Ventilators |
| • Powered Wheeled Stretcher (FDA Code: INK / 890.3690) A powered wheeled stretcher is a battery-powered table with wheels that is intended for medical purposes for use by patients who are unable to propel themselves independently and who must maintain a prone or supine position for prolonged periods because of skin ulcers or contractures (muscle contractions). |
| • Servicing, Hyperbaric/Hypobaric Chambers |
| • Ventilator Tubing and Support Set (FDA Code: BZO / 868.5975) Ventilator tubing is a device intended for use as a conduit for gases between a ventilator and a patient during ventilation of the patient. |
| • Wheeled (Mobile) Stretcher (FDA Code: FPO / 880.6910) A wheeled stretcher is a device consisting of a platform mounted on a wheeled frame that is designed to transport patients in a horizontal position. The device may have side rails, supports for fluid infusion equipment, and patient securement straps. The frame may be fixed or collapsible for use in an ambulance. |
(1451 potential buyers viewed listing, 398 forwarded to manufacturer's website)